[VT I Maryland, December 3, 2025 ] The U.S. Food and Drug Administration (FDA) held a public meeting on December 3 to initiate the reauthorization process for the Biologics Price Competition and Innovation Act (BsUFA) for fiscal years 2028–2032. The meeting marked the start of public comment solicitation and industry consultation, laying the groundwork for strategic guidance on biosimilar development, regulatory review, and market access.
Billion-Dollar Funding Boosts Review Efficiency and Innovation

Mike Davis, Deputy Director of the FDA’s Center for Drug Evaluation and Research (CDER), opened the meeting, emphasizing that since its implementation in 2012, BsUFA has been instrumental in expanding access to biosimilars, improving review efficiency, and safeguarding drug safety. Biosimilars have saved the U.S. healthcare system approximately $56 billion since 2015, with $20 billion in 2024 alone. Under BsUFA, the FDA has approved 78 biosimilars, establishing a predictable and stable review process that offers patients more affordable biologic options.

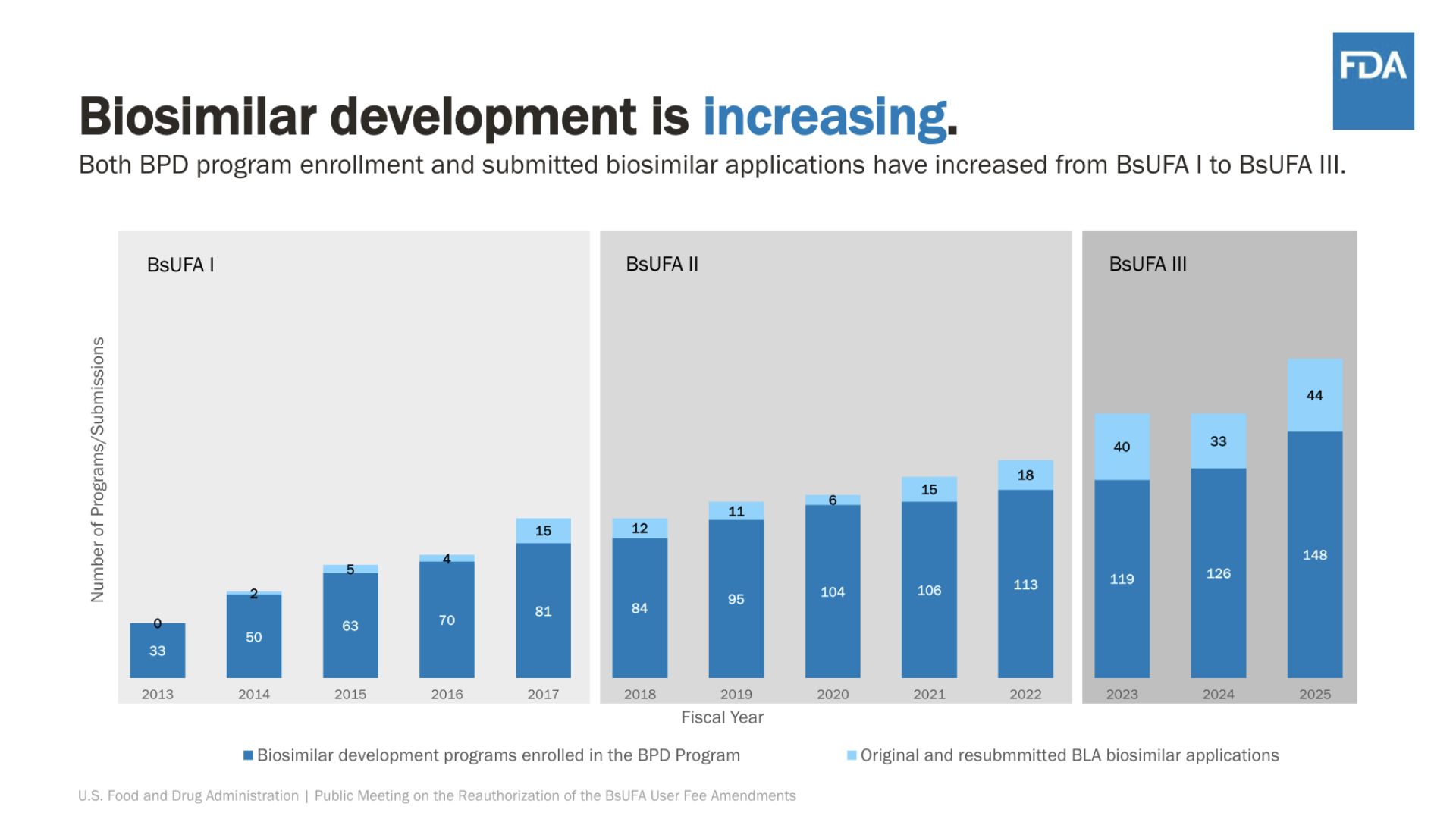
Andrew Kish, Director of CDER’s Office of Program and Strategic Analysis, outlined BsUFA’s history and three-phase development:
- BsUFA I: Established user fee structures and initial review processes, modeled after PDUFA (Prescription Drug User Fee Act).
- BsUFA II: Optimized fee structures, introduced new review commitments, and improved efficiency.
- BsUFA III: Added supplemental application types, risk analysis, and manufacturing review timelines, and launched regulatory science pilot programs to support interchangeable biosimilar development.
User fee revenue accounts for approximately 61% of the total BsUFA program budget, primarily funding review staff and process management. BsUFA III has demonstrated stable performance, improving review efficiency, meeting management, and data transparency.
Industry Highlights Review Efficiency and Market Access

Industry representatives shared expectations and recommendations for BsUFA IV.
Juliana Reed, Executive Director of The Biosimilars Forum, and Scott Kuzner, Senior Director of Sciences and Regulatory Affairs at the Biosimilars Council, emphasized that biosimilars not only lower drug costs but have saved the U.S. healthcare system billions. Key industry concerns include:
- Lengthy review timelines, especially after FDA has reduced clinical equivalence study requirements.
- The need for improved communication, transparency, and inspection efficiency.
- Support for FDA’s data-driven regulatory approach to reduce unnecessary clinical trials.
- Exploring a Single Global Comparator and optimizing human factors evaluation for drug-device combination products.
- Ensuring funding stability to avoid disruptions caused by spending trigger mechanisms.
Sean Hilscher, Senior Director of Science and Regulatory Advocacy at PhRMA, representing innovative drug companies, stressed that BsUFA IV should maintain a science-based regulatory framework, stable communication, transparent review timelines, and measurable KPIs to ensure FDA’s long-term resource stability and review predictability.
Aaron Josephson from Teva Pharmaceuticals noted that biosimilars lower costs while enhancing patient productivity and benefiting society. He recommended improvements in the review process, including better timing for information requests, reducing late-cycle labeling comments, earlier inspection scheduling, transparent use of regulatory science project funding, and assigning final approval authority to the office most familiar with the product (OTBB) to increase efficiency.
Public Engagement and Process Transparency
The meeting also marked the first step in the BsUFA reauthorization process. The public can submit written comments until January 2, 2026. Technical negotiations are expected to begin in spring 2026 and conclude by summer, followed by internal FDA review and submission of the reauthorization proposal to Congress, expected in 2027. FDA has pledged to hold at least monthly consultations with stakeholders during the process and make negotiation records publicly available to ensure transparency and traceability.
Commercial and Innovation Opportunities
For biopharmaceutical companies and startups, BsUFA IV reauthorization offers not only funding and resources but also a strategic window to accelerate approvals, optimize R&D, and expand market opportunities. Regulatory science pilot programs provide early guidance, reducing approval uncertainty, while efficient reviews and approval of interchangeable biosimilars will further open market space, creating potential benefits for innovative enterprises and investors.
The public meeting provided industry stakeholders an in-depth understanding of FDA review mechanisms and future policy directions, setting the tone for the next stage of biosimilar market development. The initiation of BsUFA IV reauthorization signals new opportunities for the U.S. biopharmaceutical industry in R&D efficiency, market competitiveness, and patient access.

The meeting was moderated by Jonathan Collins of FDA’s Center for Drug Evaluation and Research.




